Functional and Systems Biology
Engineering Enzyme Secretion in Filamentous Fungi
Technology improves enzyme’s role in biomass deconstruction for biofuel
Advanced biofuels have humble beginnings.
Inside biorefineries, plant biomass like corncobs, wood, and straw is transformed into powerful biofuels with the potential to reduce greenhouse gas emissions.
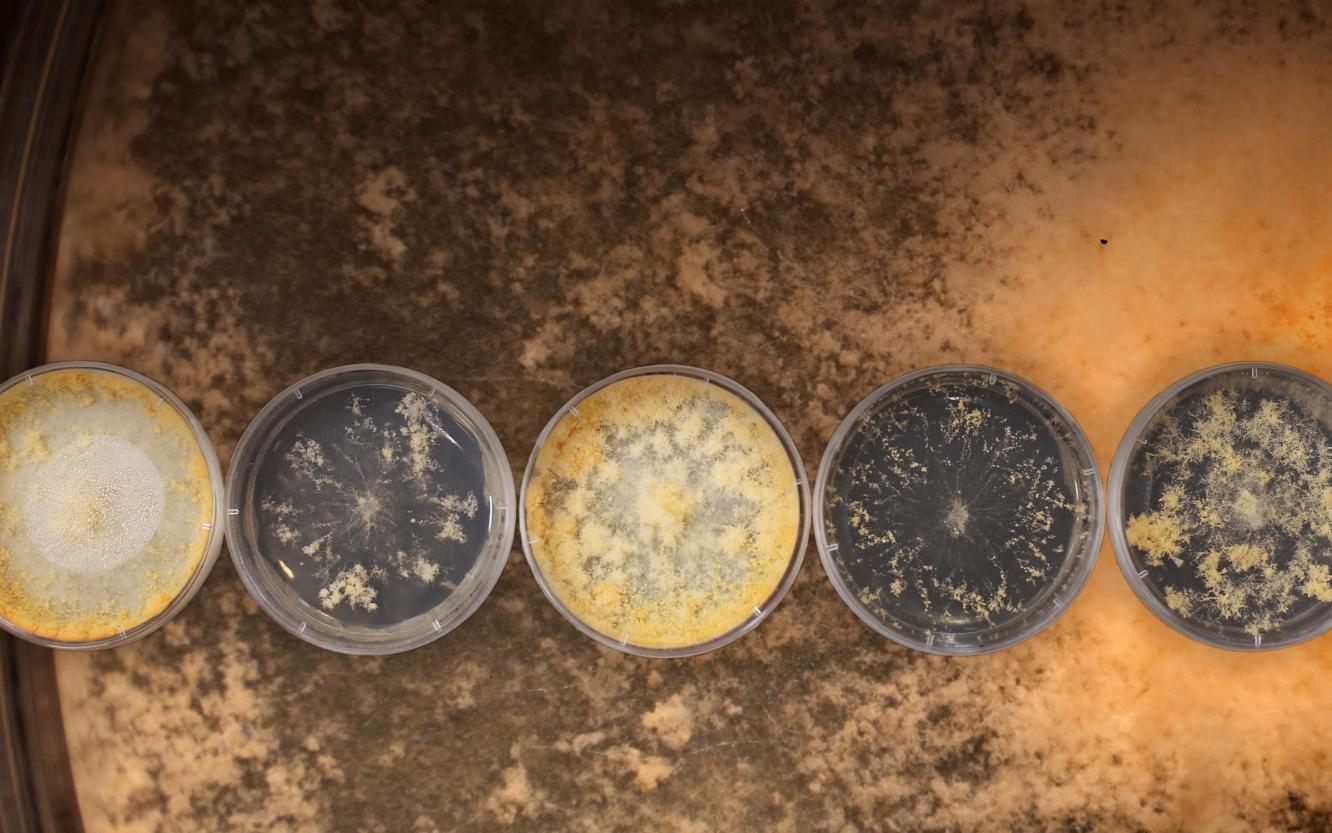
To produce biofuels, proteins known as carbohydrate active enzymes, or CAZymes, first break down the rigid plant cell walls into multiple intermediates like sugars. Traditional methods for expressing enzymes for biomass deconstruction have created a bottleneck because they’re costly, time consuming, and imprecise.
An international team of researchers, including scientists from a U.S. Department of Energy (DOE) Office of Science user facility—the Environmental Molecular Sciences Laboratory (EMSL)—and the DOE Joint BioEnergy Institute (JBEI), conducted a study to identify enzymes that could be genetically manipulated and engineered to speed up the deconstruction process. Using data from EMSL and the Joint Genome Institute (JGI)—another DOE user facility—the team identified a secretion mutant in the model fungus, Neurospora crassa, and subsequently modified the gene in a different filamentous fungi, Myceliophthora thermophila.
This modification enables the fungi to produce large quantities of enzymes. Through these systems biology and genetics studies, the research has also given insights into the regulation of enzyme secretion in filamentous fungi, allowing industrially employed fungi to be genetically engineered.
“The more we learn about the genes whose products are involved in protein secretion, the more options we have for strategies to engineer improved fungal factories for enzyme production,” said Scott Baker, one of the authors of this research. Baker leads EMSL’s Functional and Systems Biology group and has an affiliate appointment as a fungal biotechnology scientist with JBEI.
The research, “The F-box protein gene exo-1 is a target for reverse engineering enzyme hypersecretion in filamentous fungi,” was recently published in the Proceedings of the National Academy of Sciences of the United States of America.
CAZymes
For this research, the scientists used filamentous fungus known as Neurospora crassa to identify a mutation in a gene that regulates the production of CAZymes. Filamentous fungi are important sources of CAZymes as well as cell factories for diverse biotechnological products, including enzymes. Filamentous fungi also are known for their “unparalleled secretion capacity.” The researchers applied information from the model biological system, Neurospora crassa, to engineer an industrially relevant organism known as Myceliophthora thermophila, which has CAZymes.
Genome sequencing
Using a technique known as genome sequencing, the team mapped all of the mutations in Neurospora crassa, then identified the specific mutation causing the hypersecretion phenotype and performed an analysis to see how it regulated the production of CAZymes. After the team confirmed that the N. crassa mutation caused the CAZyme hypersecretion, they used a gene editing technology called CRISPR Cas-9 to delete the orthologous gene, or the gene that separated from Myceliophthora thermophila. Orthologous genes evolve from a common ancestor gene and maintain similar function in different organisms.Next, the team tested the modified fungus, confirming it secreted higher levels of CAZymes.
Bioenergy and beyond
This research demonstrates how classical mutants and next-generation sequencing can uncover unsuspected candidates for engineering. Data from this research contributes to a more complete understanding of CAZyme regulation and facilitates targeted engineering of hypersecretion in other organisms.
“Understanding this process will improve our ability to transition to a green-carbon economy where energy biomass is recycled, not just harvested,” said Kevin McCluskey, a co-author and principal scientist at Bolt Threads.

